- Home
- Search
- Images
- Species Checklists
- US States: O-Z >
- US National Parks
- Central America
- South America
- US National Parks
- Southern Subpolar Region
|
|
|
|
Family: Physciaceae
|
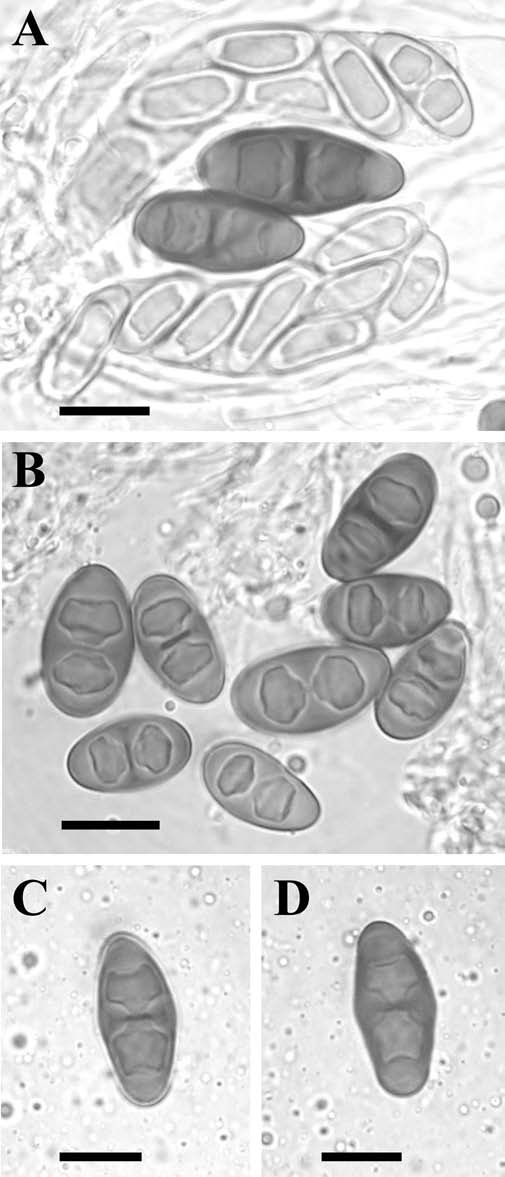
MB#561626 Type. CANADA. ALBERTA. 1 mile SE Gap, 115.15 W 51.03 N, Populus tremuloides ‑ P. balsamifera woods, 4 250 ft, on P. balsamifera, 7 June 1972, R.A. Kalgutkar 1169 (PMAE ‑ holotype). Description. Thallus thin, light or dark grey, discontinuous, scabrid in appearance, comprised of discrete areoles to ca. 0.20 mm wide, sometimes coalescing to form a continuous or rimose thallus; surface plane to mostly rugose, matt; margin indeterminate; prothallus absent; vegetative propagules present; papillate blastidia frequent, 50-100 x 100-150 µm, and/or marginal consoredia spreading to surface 30-35 µm in diam., breaking into soredia 10-25 µm in diam., all propagules darker than thallus. Apothecia broadly attached to narrowly attached, frequent, contiguous or not, to 0.35‑0.70 mm diam.; disc dark brown to black, plane, sometimes becoming markedly convex; margin concolourous with thallus, 0.05‑0.10 mm wide, entire, persistent; excipular ring absent. Apothecial Anatomy. Thalline exciple 50‑100 µm wide laterally; cortex cellular, 10‑20 µm wide laterally, often with short hyphal outgrowths; epinecral layer sometimes present, <5 µm wide; crystals absent in cortex and medulla; cortical cells to 4.0‑7.0 µm wide, pigmented or not; algal cells to 10.5‑15.0 µm long; thalline exciple 60‑120 µm deep below; cortex 10‑40 µm wide; proper exciple 5‑10 µm wide laterally, to 20‑40(-60) µm at periphery, usually hyaline but sometimes lightly pigmented like hypothecium; hypothecium hyaline or yellowish, 40‑80 µm deep; hymenium 70‑110 µm high, not inspersed; paraphyses 2.0‑3.0 µm wide, not conglutinate, apices to 4.0‑6.5 µm, lightly pigmented forming a light to reddish brown epihymenium; asci 60‑80 x 15‑22 µm. Ascospores 4 to mostly 8/ascus, Type B development, Dirinaria‑type (Figure 9), (15.0-)19.0‑20.0(‑24.0) x (6.5‑) 9.0-10.0(‑13.0) µm, average l/b ratio 1.9-2.2, mostly inflated at septum, more so in KOH, lumina angular at first (Physcia-like) becoming more rounded, retaining thick apical walls, often with septal disc at maturity; torus lacking; walls lightly ornamented or not. Pycnidia spherical, surficial, pigmented brown; conidiophores Type I or V; conidia bacilliform, 3.5-5.0 x 1.0 µm. Chemistry. Spot tests all negative; secondary metabolites not tested. Substrate and Ecology. Corticolous, collected on Fraxinus, Juniperus, Populus angustifolia, P. balsamifera, P. tremuloides, Pinus flexilis, and Salix primarily in riverine habitats, recorded from elevations of 720-1 880 m in the west. Also on Acer saccharum in Michigan. These and the Minnesota collections were accompanied by R. subminuta. Distribution. A North American endemic species distributed in the Rocky Mountain foothills from Alberta to Colorado, east into the Dakotas, with outliers in Minnesota and Michigan. Notes. Rinodina albertana is characterized by its Dirinaria-type spores and scabrid thalli in the western part of its distribution, with the frequent presence of papillate blastidia and/or consoredia. The vegetative propagules are darker than the thallus and may give the species a superficial resemblance to R. colobina in the field. The Minnesota and Michigan specimens have better developed blastidia which are not dark in colour, and no asci with a reduced number of spores were observed in this population. Another notable feature is the production of fewer than eight spores by some asci. Eight ascospores are initially differentiated but four may fail to develop to maturity. The spores are variable in size (CV >10% for both length and breadth) and structure, a common feature of species with vegetative propagules in Rinodina. Another distinguishing feature of some specimens is the production of short hyphae by the cortical cells which may give an hirsute appearance to the surface of the thallus and apothecial margins. Despite the many characteristics listed above, this species may easily overlooked because of the small size and poor development of the thalli. The spores are similar to R. metaboliza, but the average spore size of R. albertana is significantly larger. Rinodina riparia is characterized by larger spores and a better developed thallus which lacks vegetative propagules. This species has a more southerly centre of distribution and does not extend north into Canada. The Minnesota specimen of R. albertana was previously included in R. thomsonii (Sheard 1995). The remaining specimens listed under this name, including the type, are now included in R. santae-monicae. This last species rarely possesses vegetative propagules that then tend to be larger than those of R. albertana. In addition, R. santae-monicae has a much less conspicuous Type B spore development and has a Pacific coast distribution. Specimens examined. CANADA. ALBERTA. 11 mi N Jasper, W.A. Weber 23419 (COLO); 16 mi N Lundbreck, R.M. Kalgutkar 2449; 17 mi N Lundbreck, R.M. Kalgutkar 2552; Edson, R.M. Kalgutkar 1002; Exshaw, R.M. Kalgutkar 1200, 1237, 1250, 1285, 1290; Gap, R.M. Kalgutkar 1169, 1195, 1197; Grotto Mtn., 1968, C.D. Bird 21665 (all PMAE); Thompson Creek Campground, J.W. Sheard 5267c (SASK). U.S.A. COLORADO. Boulder Co., Gregory Canyon, J.W. Sheard 4660 (SASK); Jefferson Co., Deer Creek, 1973, M‑B. Carmer (SASK); M-B. Carmer 1710 (COLM, COLO, BRY). MICHIGAN. Alger Co., Pictured Rocks Nat. Lakeshore, C.M. Wetmore 58766, 58779. MINNESOTA. St. Louis Co., 11 mi SE Aurora, C.M. Wetmore 30851. NORTH DAKOTA. Billings Co., 1 mi N Medora, C.M. Wetmore 45516; 6.5 mi N Medora, C.M. Wetmore 80399 (all MIN). SOUTH DAKOTA. Pennington Co., edge of Sheep Mtn., S. Will-Wolf 2070; Shannon Co., Fog Creek drainage, S. Will-Wolf 2817 (both WIS). |