Consortium of Lichen Herbaria
- building a Global Consortium of Bryophytes and Lichens as keystones of cryptobiotic communities -
- Home
- Search
- Images
- Species Checklists
- US States: O-Z >
- US National Parks
- Central America
- South America
- US National Parks
- Southern Subpolar Region
|
|
|
|
Family: Caliciaceae
[Buellia chloroleuca Körb., moreBuellia poeltii T. Schauer] |
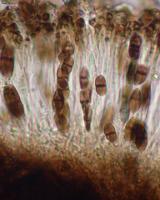
Nash, T.H., Ryan, B.D., Gries, C., Bungartz, F., (eds.) 2007. Lichen Flora of the Greater Sonoran Desert Region. Vol 3. Thallus: crustose, granular areolate, moderately thickened, ±continuous or becoming dispersed; prothallus: absent or delimiting the thallus as a black outline where several different thalli meet surface: dark to pale gray, fresh material is often greenish, fading to brownish gray in storage, dull, slightly roughened, epruinose, phenocorticate, esorediate medulla: white, lacking calcium oxalate (H2SO4-) Apothecia: lecideine; (0.2-)0.3-0.7(-0.9) mm in diam., soon sessile margin: black, thin, rarely persistent, soon excluded, rarely with a thalline veil when emerging from the thallus disc: black, epruinose, plane, soon becoming convex proper exciple: narrow, poorly differentiated, aethaleatype, inner excipular hyphae narrow, hyaline, prosoplectenchymatous (textura oblita), often reduced, similar in structure and orientation to the paraphyses, transient with the deep reddish brown hypothecium (leptoclinoidesbrown, textura intricata), outer excipular hyphae parallel, moderately swollen (textura oblita) and usually strongly carbonized with various amounts of a brown pigment (cf. elachista-brown, HNO3-) epihymenium: brown, pigmentation continuous with the outer exciple (HNO3-) hymenium: hyaline, not inspersed with oil droplets, but sparse oil droplets present from a weakly inspersed subhymenium; paraphyses: simple to moderately branched, apically swollen, with a brown pigment cap (cf. elachista-brown) asci: clavate, Bacidia-type, 8-spored ascospores: soon brown, 1-septate, occasionally with two additional false septa, narrowly ellipsoid, not constricted, with ±tapered ends, frequently slightly curved, (10.5-)16-[18.8]-21.7(-27) x (5.6-)7.2-[8.3]-9.4(11.2) µm (n=76); proper septum: narrow, barely thickening during spore ontogeny, lateral wall inconspicuously thickened (±Callispora-type); ornamentation: faintly microrugulate (barely seen without DIC) Pycnidia: rare, globose, unilocular; ontogeny similar to the Umbilicaria-type conidiogenous cells: mostly terminal, rarely also intercalary (cf. conidiophore-type V) conidia: bacilliform, 3-6.5 x 0.5-1 µm (n=80) Spot tests: K+ yellow, P-, C+ distinctly or weakly orange or C- fluorescence: UV- (pale) or + weakly orange iodine reaction: medulla non-amyloid Secondary metabolites: 6-O-methylarthothelin, ±arthothelin with isoarthothelin, ±atranorin, occasionally also 4,5-dichloro-6-O-methylnorlichexanthone, 5,7-dichloro-3-O-methylnorlichexanthone, 5,7-dichloro-3-O-methylnorlichexanthone, 5,7-dichloronorlichexanthone, brialmontin 1, variolaric acid (J. A. Elix, HPLC). Substrate and ecology: on a variety of mostly coniferous trees like Abies, Pseudotsuga, or Pinus, also on Quercus, frequently at the tree bases on ±decaying wood or bark, in montane and subalpine forests, usually above 2000 m World distribution: widely distributed throughout the USA and Europe Sonoran distribution: widely distributed in Arizona. Notes: Reports of the recently described Amandinea subduplicata Marbach for the Sonoran Desert Region are based on misidentifications and belong instead to Buellia chloroleuca. Another specimen from the Sonoran Region identified by Marbach as A. submontana also belongs to B. chloroleuca. Marbach (2000) did not discuss B. chloroleuca and generally did not give C-reactions for any specimens examined. Sonoran specimens reported as either one of these two species by Marbach all have bacilliform conidia. Some of the specimens distinctly react C+ orange and thus contain xanthones. One specimen mentioned by Marbach does not contain xanthones, but it is also characterized by bacilliform conidia. This specimen also has different ascospores and is placed in B. erubescens. Most specimens of B. chloroleuca are very distinct and easily recognized by their granular thalli and conspicuous C+ orange reactions. Nevertheless, the species has not previously been reported from North America (Esslinger 1997). When Giralt et al. (2000) published their treatment on some European corticolous Buellia species, they pointed out that Schauer (1965) had not recognized the synonymy of B. zahlbruckneri with B. erubescens due to confusion of type material. Schauer (1965) even described a new species as B. poeltii, which must now be regarded as a synonym of B. chloroleuca (Giralt et al. 2000). North American lichenologists subsequently treated B. zahlbruckneri as a synonym of B. erubescens, even though most North American material identified as B. zahlbruckneri sensu Imshaug (1951) clearly belongs to B. chloroleuca. Both species have comparatively narrow spores with distinct tendencies towards the Callispora-type. In contrast, B. erubescens has broadly ellipsoid spores. Imshaug (1951) must have been aware of the differences when he discussed B. zahlbruckneri in the context of B. disciformis, also a species with Callispora-type ascospores. Giralt et al. (2000) emphasized that a C spot test reaction of the thallus may not always be reliable and instead emphasized the UV+ orange fluorescence caused by the xanthones in the thallus. In Sonoran material this UV reaction is typically weak and barely noticeable; in contrast, the Sonoran material is often distinctly C+ orange, especially if thallus sections are tested in the compound microscope. Specimens without a distinct reaction were initially believed to belong to a different species, but using HPLC traces of xanthones were also detected in these specimens. |
|
|
|
Powered by Symbiota